Mori, L., De Libero, G. ‘Bohemian Rhapsody’ of MR1T cells. Nat Immunol 21, 108–110 (2020).
An exciting new finding in immunology offers hope for a brand new cancer treatment in the future. In an article published last week in Nature Immunology Journal, researchers from Cardiff University reported finding a new T- cell receptor which recognizes most human cancer types and activates immune responses against them, while ignoring healthy cells.
T-cell therapies are the newest approach to cancer – using lymphocytes (white blood cells) drawn from the patient and potentized or activated before being put back to mount a defense against cancer. However, side effects are unacceptably high and, so far, they can only target a few types of cancer.
The hope is that this new T-cell treatment will be better tolerated, have wide application and translate into a whole new approach to treating cancer.
With a herbal medicine practice focused on holistic oncology, I am always interested in the latest ground breaking medical research. New findings may take 10 years or more to filter down to general practice, but the clinical approach to cancer care has changed dramatically in the past few years and certainly the outlook for many patients is more positive today then it ever was in the past.
When I started in this specialty almost 20 years ago, it was challenging to even get Her2/neu receptor testing in breast cancer patients. Now this is standard practice and it would be considered negligent to miss doing it. The drug Herceptin targets this site specifically, and since it came to market there are tens of dozens more of these targeted immunotherapy drugs that are either now available or in the pipeline. They are effective and exciting if you exhibit the particular genetic mutations that make your cancer a target for the drug, and they are generally better tolerated than conventional chemotherapy, although of course they’re not without side effects and consequences.
For the herbalist the challenge is to provide the best possible protocol to work alongside the drugs and to assist them to be more effective and have less side effects. For example, taking high dose green tea, and especially the EgCG compound found in it, will potentize and augment Herceptin.
Herbs and the immunotherapies
‘The new immunotherapy drugs, especially the T cell therapies, are all aiming to use the patients own immune system to fight back against their cancer, so this is a great opportunity to the immune enhancing / immune tonic herbs. The complex polysaccharides (large sugars) and saponins in Astragalus membranaceous (milk vetch) and other sugars and saponins in various medicinal mushrooms, are used for this, often in the form of a ‘blood building’ soup. This may include bone marrow, root vegetables, stinging nettle leaf, green leafy vegetables, seaweeds, beans, and good sources of protein (red meat, fish).
Take Echinacea tincture 1 tsp (5 mL) three times daily for 2 days after Immunotherapy treatments
Take a sauna or a very hot bath the day of and the day after receiving the immunotherapy drugs, and drinking 3 cups daily of Interferon Inducing tea.
Interferon Inducing tea for 2 days after Immunotherapy treatments
Elderflower, Elderberry, Yarrow flower, Boneset, Peppermint. 50 g of each, mixed
Take a knob of fresh ginger about 1” long and chop fine. Put in a pan with 1 L cold water. Cover and bring to a boil then turn right down and simmer on low for 5 minutes. Pour over 6 tsp. herb mix, steep 15 minutes. Sweeten to taste with honey.
These suggestions are useful for cancer patients receiving immunotherapies but also for anyone who needs to mount a sudden and powerful immune system response to thwart an infection, especially if there is fever.
Below is a lengthy article outlining how the therapy works and why it may be ground breaking in cancer care.
https://www.goodnewsnetwork.org/new-t-cell-could-lead-to-universal-cancer-therapy
New type of killer T-cell that offers hope of a universal cancer therapy.
Researchers at Cardiff University have discovered a new type of killer T-cell that offers hope of a “one-size-fits-all” cancer therapy.
T-cell therapies for cancer—where immune cells are removed, modified and returned to the patient’s blood to seek and destroy cancer cells—are the latest paradigm in cancer treatments.
The most widely-used therapy, known as CAR-T, is personalized to each patient, but it only targets a few types of cancers and has not been successful for solid tumors, which make up the vast majority of cancers.
Cardiff researchers have now discovered T-cells equipped with a new type of T-cell receptor (TCR) which recognizes and kills most human cancer types, while ignoring healthy cells.
This TCR recognizes a molecule present on the surface of a wide range of cancer cells as well as in many of the body’s normal cells but, remarkably, is able to distinguish between healthy cells and cancerous ones, killing only the latter.
The researchers said this meant it offered “exciting opportunities for pan-cancer, pan-population” immunotherapies not previously thought possible.
How does this new TCR work?
Conventional T-cells scan the surface of other cells to find anomalies and eliminate cancerous cells—which express abnormal proteins—but ignore cells that contain only “normal” proteins.
The scanning system recognizes small parts of cellular proteins that are bound to cell-surface molecules called human leukocyte antigen (HLA), allowing killer T-cells to see what’s occurring inside cells by scanning their surface.
HLA varies widely between individuals, which has previously prevented scientists from creating a single T-cell-based treatment that targets most cancers in all people.
But the Cardiff study, published this week in Nature Immunology, describes a unique TCR that can recognize many types of cancer via a single HLA-like molecule called MR1.
Unlike HLA, MR1 does not vary in the human population—meaning it is a hugely attractive new target for immunotherapies.
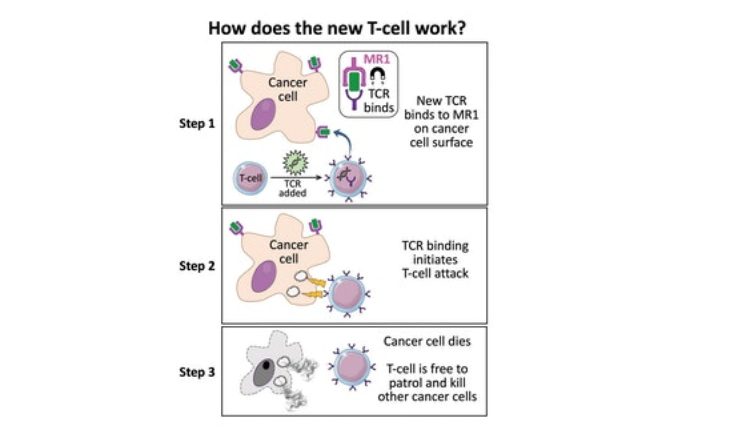
What did the researchers show?
T-cells equipped with the new TCR were shown, in the lab, to kill lung, skin, blood, colon, breast, bone, prostate, ovarian, kidney and cervical cancer cells, while ignoring healthy cells.
To test the therapeutic potential of these cells in vivo, the researchers injected T-cells able to recognize MR1 into mice bearing human cancer and with a human immune system.
This showed “encouraging” cancer-clearing results which the researchers said was comparable to the now NHS-approved CAR-T therapy in a similar animal model.
The Cardiff group were further able to show that T-cells of melanoma patients modified to express this new TCR could destroy not only the patient’s own cancer cells, but also other patients’ cancer cells in the laboratory, regardless of the patient’s HLA type.
Professor Andrew Sewell, lead author on the study and an expert in T-cells from Cardiff University’s School of Medicine, said it was “highly unusual” to find a TCR with such broad cancer specificity and this raised the prospect of “universal” cancer therapy.
“We hope this new TCR may provide us with a different route to target and destroy a wide range of cancers in all individuals,” he said.
“Current TCR-based therapies can only be used in a minority of patients with a minority of cancers. Cancer-targeting via MR1-restricted T-cells is an exciting new frontier – it raises the prospect of a ‘one-size-fits-all’ cancer treatment; a single type of T-cell that could be capable of destroying many different types of cancers across the population. Previously nobody believed this could be possible.”
What happens next?
Experiments are under way to determine the precise molecular mechanism by which the new TCR distinguishes between healthy cells and cancer. The researchers believe it may work by sensing changes in cellular metabolism which causes different metabolic intermediates to be presented at the cancer cell surface by MR1. The Cardiff group hope to trial this new approach in patients towards the end of this year following further safety testing.
Professor Sewell said a vital aspect of this ongoing safety testing was to further ensure killer T-cells modified with the new TCR recognize cancer cells only.
“There are plenty of hurdles to overcome however if this testing is successful, then I would hope this new treatment could be in use in patients in a few years’ time,” he said.
Professor Oliver Ottmann, Cardiff University’s Head of Haematology, whose department delivers CAR-T therapy, said: “This new type of T-cell therapy has enormous potential to overcome current limitations of CAR-T, which has been struggling to identify suitable and safe targets for more than a few cancer types.”
Professor Awen Gallimore, of the University’s division of infection and immunity and cancer immunology lead for the Wales Cancer Research Centre, said: “If this transformative new finding holds up, it will lay the foundation for a ‘universal’ T-cell medicine, mitigating against the tremendous costs associated with the identification, generation and manufacture of personalized T-cells.
“This is truly exciting and potentially a great step forward for the accessibility of cancer immunotherapy.”